|
|
Information box |
The main purpose of this site is to extend the
intraoperative monitoring to include the neurophysiologic
parameters with intraoperative navigation guided with Skyra 3
tesla MRI and other radiologic facilities to merge the
morphologic and histochemical data in concordance with the
functional data.
 CNS Clinic
CNS Clinic
Located in Jordan Amman near Al-Shmaisani hospital, where all
ambulatory activity is going on.
Contact: Tel: +96265677695, +96265677694.
 Skyra running
Skyra running
A magnetom Skyra 3 tesla MRI with all clinical applications
started to run in our hospital in 28-October-2013.
 Shmaisani hospital
Shmaisani hospital
The hospital where the project is located and running diagnostic
and surgical activity. |
|
 |
|
 |
 |
Echo-planar
imaging (EPI) and functional MRI |
 |
1. Introduction
Since the first days of human NMR imaging, reaching back
to the late 1970’s, imaging time has presented a serious
practical limitation. The practical reality of ordinary
structural imaging is that normal subjects are willing
to tolerate perhaps an hour of lying inside of the
imaging magnet, and are able to stay still for little
more than fifteen minutes. Both NMR contrast and signal
to noise ratio, however, are time-dependent phenomena.
As a result, imaging time and image quality have
traditionally been at odds for all manner of magnetic
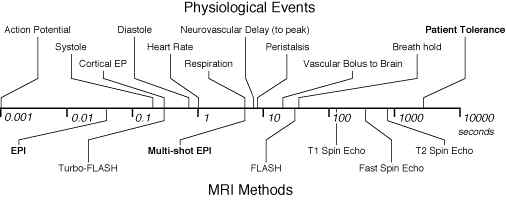
resonance imaging. Figure 1 shows the relationship
between imaging time and a variety of interesting
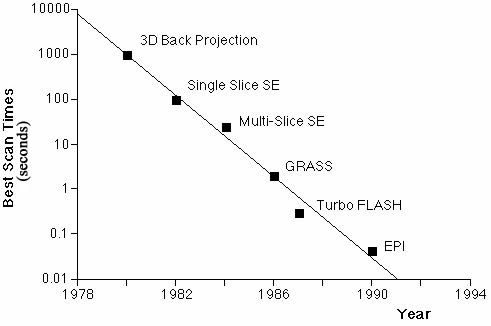
biological phenomena. Figure 1B, shows the steady
decrease in practical MR imaging times that took place
over the first decade of human imaging and demonstrates
the remarkably steady logarithmic improvements in
imaging speed that have characterized the field. It is
perhaps even more remarkable, therefore, that today’s
fastest practical imaging method, echo-planar imaging,
or EPI, was conceived in 1977, before the veritable
explosion in clinical use of MRI. EPI achieved largely
novelty status however, until it found a driving
application – namely functional MR imaging and
especially functional neuroimaging. A major factor in
the relatively slow acceptance of EPI is that it is just
plain hard to implement as we will see below.
|
 |
 |
|
A |
B |
Figure 1A. Comparison of physiological processes and
imaging speeds of common magnetic resonance imaging
methods. To avoid image artifacts, scan times must not
be longer than the duration of motion. To study the
dynamics of these processes, the imaging times must be
substantially shorter. EPI and multi-shot EPI, are indicated in
bold. B. Fastest MRI scan times as a function of year and technology.
2. What is EPI?
While MRI as conventionally practiced builds up the data
for an image from a series of discrete signal samples,
EPI is a method to form a complete image from a single
data sample, or a single "shot". The speed advantages
can be astonishing. For example, a typical T2-weighted
imaging series (to form an image whose contrast depends
predominantly on the intrinsic tissue magnetization
parameter, T2) requires that the time between excitation
pulses, known as "TR" be two to three times longer than
the intrinsic tissue magnetization parameter, T1. The T1
of biological samples is typically on the order of a
second or so (cerebrospinal fluid, or CSF, can have much
longer T1’s of several seconds); TR must therefore be 3
seconds or more. A more or less typical MR image is
formed from 128 repeated samples, so that the imaging
time for our canonical T2 weighted scan is about 384
seconds, or more than 6.5 minutes. By comparison, the
EPI approach collects all of the image data, for an
image of the same resolution, in 40 to 150 milliseconds
(depending on hardware and contrast considerations).
This reflects a nearly 10,000-fold speed gain.
Although, there are myriad
variations, EPI is fundamentally just a trick of spatial
encoding. To understand the difference between EPI and
conventional imaging, it is necessary therefore, to have
some understanding of spatial encoding in MRI.
2.1 MRI Spatial Encoding
Tomographic image formation requires spatial encoding in
three dimensions. In most cases, one dimension is
determined by slice selective excitation (refer to
Figure 2 for axis labels). Briefly, a radio frequency
excitation pulse with a narrow frequency range is
transmitted to the subject in the presence of a spatial
magnetic field gradient. Because the magnetic resonance
phenomenon depends on an exact match between the radio
frequency excitation pulse frequency and the proton spin
frequency, which depends in turn, on the local magnetic
field, this pulse will excite the MR signal over a
correspondingly narrow range of locations: an imaging
slice. The differences between EPI and conventional
imaging occur in the remaining "in-plane" spatial
encoding.
|
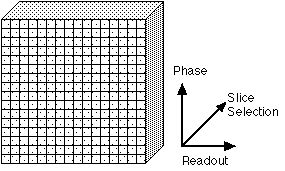 |
Figure 2. The three axes used for spatial encoding of MR
images. One dimension of spatial encoding is achieved by
slice selective excitation (the "Slice Selection" axis).
The other two are encoded by phase and frequency. Some
texts will refer to the Slice Selection axis as the "Z"
axis. The Readout axis is variously labeled the
"Frequency" or "X" axis; the Phase Encoding axis is
sometimes labeled the "Y" axis.
When a magnetic field gradient is applied across this
excited slice, it will cause the spin frequency to be a
function of position. The pixel size, or spatial
resolution, of an MR image depends on the product
(actually the integral) of the imaging gradient
amplitudes and their ON duration. Specifically, the
pixel size is equal to 1/gGt, where g is the Larmor
constant (4258 Hz/gauss), G is the gradient amplitude,
usually expressed in gauss/cm, and t is the gradient on
time. A gradient of 0.5 gauss/cm, left on for 10 msec,
for example, yields a spatial resolution of 0.47 mm.
This, however, reflects spatial encoding along one
in-plane dimension only – the "Readout" direction. In
ordinary two-dimensional Fourier transform imaging, the
encoding for the second in-plane dimension is created by
applying a brief gradient pulse (along a second gradient
axis) before each readout line. For 128 lines of
resolution in this axis, 128 separate lines must be
acquired, each for 10 msec. The total readout duration
is therefore 128 x 10 msec, or 1.28 seconds.
Unfortunately, the MR signal lasts for only about 100
milliseconds (limited by T2) and over the course of a
1.28 second readout duration (spatial encoding period)
the signal will have decayed to nothing.
In EPI, much larger gradient amplitudes are used. A
gradient of about 2.5 gauss/cm is typical, but human
imagers with gradient amplitudes in excess of 5 gauss/cm
are achievable. With five times the gradient amplitude,
the encoding duration can be reduced by five-fold, to 2
msec/line, so that the total spatial encoding time for
our reference image is reduced from 1.28 seconds to 256
msec. The human brain has a T2 of about 100 msec at
typical imaging field strengths. Thus, a 256 msec
readout might be marginally realistic. In practice,
however, this is not a
practical configuration. Most significantly, the
gradients cannot instantly reach such large magnitudes,
and the rise time therefore becomes a significant
fraction of the readout duration. Secondly, the decay of
the MR signal during readout introduces blurring into
the images. Because of these tradeoffs, most EPI
studies are performed at somewhat lower resolution.
In-plane voxel sizes between 1.5 and 3 mm are typical.
In many cases all of this is somewhat easier to
understand in terms of "k-space", where k-space is a
representation of the MRI raw data before it has been
Fourier-transformed in order to make an image.
The signal location in k-space is the integral of the
gradient amplitude and on-time:
ki(t) =
ϒ ∫Gi(t)dt
where ki is the location in k-space along the i axis,
Gi(t) is the gradient amplitude along the i axis as a
function of time, g is the Larmor constant, and t is the
gradient on-time. As the gradient-time product
increases, that is, as the signal is encoded to higher k
values, the image resolution increases. Thus, in order
to make an MR image of any desired final resolution, we
must collect MR data over a corresponding area of
k-space.
In conventional MRI, k-space is covered line by line, as
suggested in figure 3A. Following each RF excitation, a
single line of raw data is collected along kx (the
readout axis), with sequential lines acquired at
different displacement along the ky axis. Because a
separate excitation step is required prior to the
collection of each data line, the total imaging time
depends on the time between excitations (also known as
tr) as well as on the total number of data lines
collected. The latter depend on the desired resolution
and field of view in the final images.
|
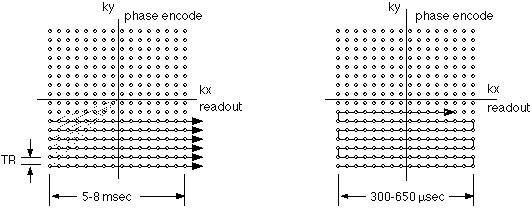 |
Figure 3A and 3B. K-space traversal patterns used in
conventional imaging (A. left) and echo-planar imaging
(B. right). Small circles represent the required data
samples. In conventional imaging, each raw data line is
separately acquired after an RF excitation. As a result,
tr elapses between the collection of each data line. In
EPI, the lines are acquired continuously, in a
raster-like pattern, with as little as 300 µsec elapsing
from line to line.
Figure 3B shows the k-space trajectory used in
echo-planar imaging. Here, the sequential raw data lines
are acquired immediately after one another. In modern
imagers, the collection of each data line can be as
rapid as 300 microseconds. Figure 4 shows the gradient
encoding scheme needed for the EPI k-space trajectory.
The rapid back and forth traversal of the readout axis
is preformed by using an oscillating readout gradient.
Following each readout excursion, a brief pulse of the
phase encoding gradient is used to move to the next line
in the phase encode direction.
|
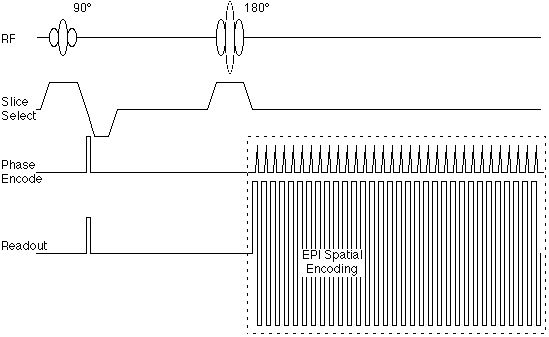 |
Figure 4. Echo-planar pulse sequence corresponding to
the k-space trajectory shown in figure 3B.
Figure 4 suggests also that the echo-planar encoding
portion of the sequence can be encapsulated as a module
that is somewhat independent of the radio frequency
pulse sequence. This is important,
as the image contrast is determined largely by the RF
sequence, rather than the gradient spatial encoding.
3. SNR
Signal to Noise ratio (SNR) in MRI is a function of:
-
Available transverse magnetization (pulse sequence and
contrast)
-
Imaging time (or more precisely, time spent receiving
the signal)
-
Bandwidth – the signal sampling rate
-
Field Strength
-
RF coil loading, coupling and sensitivity.
-
Voxel volume
Echo planar imaging carries the advantage (by contrast
with the so-called gradient echo methods such as FLASH,
GRASS, etc…) that the full magnetization is
available as signal: In a single shot method, all of the
longitudinal magnetization may be used in image
formation without a penalty in overall imaging time. The
imaging time consideration also works to the advantage
of EPI: in a typical EPI sequence, as used in ƒMRI,
signal may be collected for well over 75% of the imaging
time, leading to a very high efficiency. EPI pays a
penalty in bandwidth, however. The imaging speed in EPI
comes from the use of very high amplitude field
gradients which, in turn allow, and require, very rapid
sampling. While conventional MR imaging may use receiver
bandwidths up to about 32 kHz, bandwidths of 300 kHz are
typical in EPI and drop the usable SNR by about
two-thirds. Field strength, and RF coil considerations
are not pulse-sequence dependent. All told, theoretical
analyses and direct measurement have demonstrated a
nearly five-fold SNR advantage of EPI over FLASH studies for comparable voxel volume.
4. Bandwidth and Artifacts
The bandwidth of an MR image refers the difference in MR
frequencies between adjacent pixels, as well as to the
total range of frequencies that make up an image. In
conventional imaging , the bandwidth, per pixel, is
ordinarily kept comparable to the chemical shift between
fat and water. In a 1.5 Tesla instrument, for example, a
pixel bandwidth of 125 Hz is typical (the fat-water
shift is about 220 Hz). In this case, the fat and water
components of a single voxel will be shifted from one
another by about 1 pixel, which is an acceptable imaging
artifact. At first blush, one would expect that the
pixel bandwidth in EPI would be very high, due to the
rapid sampling rate. This is, in fact, true along the
readout axis. The continuous encoding scheme used in
EPI, however, results in a relatively low bandwidth
along the phase encoding axis; 30 Hz/pixel is typical.
This causes several difficult artifacts to occur in EPI.
4.1 Chemical Shift
In echo planar imaging, the very low bandwidth along the
phase encode axis results in substantial chemical shift
artifacts. At 1.5 Tesla, for example, using a 30
Hz/pixel bandwidth, fat and water are displaced by about
8 pixels. Further, the voxel sizes in EPI are usually
rather large, for the reasons discussed above. Using a
more or less typical 3 mm voxel, fat and water may be
displaced from one another by 2.5 cm. This problems
scales with field strength, so that in a 3 Tesla scanner
the fat water chemical shift approaches 5 cm. Since most
body tissues contain at least some water and fat, it is
absolutely necessary to correct for the chemical shift
problems.
Fortunately, there are a number of good technologies to
manage chemical shift. In the vast majority of
cases, only the water component of the MR signal is of
clinical interest. This is always the case in functional
neuroimaging, where the lipid content of the brain is
very low and the dominant source of fat signal is the
component found in skin. It is therefore reasonable to
simply suppress the fat signal outright. Usually, this
is done by applying a fat saturation pulse prior to
imaging. Because the chemical shift between fat and
water is quite large, one can transmit a 90° pulse at
the fat frequency without significantly affecting the
water signal. After this pulse the fat signal will be in
the transverse plane and it can be dephased easily by
applying a gradient pulse. Until the fat signal has had
time enough to recover its longitudinal magnetization it
will not appear in the images. This so-called chemical
shift saturation method does require excellent magnetic
field homogeneity so that the frequencies of fat and
water are well-resolved. Fortunately, today’s imaging
instruments easily meet this requirement.
An alternative method of suppression is to use STIR
(short TI inversion recovery). This approach takes
advantage of the T1 difference between fat and other
body tissues. An inversion (180°) pulse is applied
immediately prior to the EPI imaging sequence timed such
that the magnetization of fat is recovering through zero
at the time of the 90° excitation pulse. Because the fat
has no magnetization at that time, the 90° pulse does
not result in the formation of any signal from fat.
While STIR is a very effective method of fat
suppression, it has side effects that make it less
desirable. First of all, it alters the contrast of the
images overall, as it adds T1 contrast. Secondly, the
method works best if the inversion pulse is applied only
when the tissue is fully magnetized. The latter requires
that inversion recovery be used only with long TR
images.
4.2 Shape Distortion
The
low bandwidth of EPI causes a much less manageable artifact in
shape distortion. Even in a well-shimmed magnet, the human head
will magnetize unevenly so that the MR frequency may differ from
point to point by about 1 part per million (ppm). These small
frequency differences result in spatial displacement of the
signal in the resulting images. Most investigators simply
tolerate the typical one or two pixel distortion as an
acceptable artifact. Generally, however, this artifact is
correctable. It is possible to measure the magnetic field in the
head and then to apply a correction to the MR image to shift the
signal to its correct location.
The shape distortions are a frequent cause of concern in
functional neuroimaging, as it is often desirable to
superimpose regions of brain activation onto higher
resolution structural images, that are usually acquired
conventionally (e.g., with a much higher bandwidth). In
this case, the activation maps will not be registered
properly with the structural data set.
4.3 Ghosting
When the MR field gradients are switched on and off, the
time varying magnetic field of the gradients results in
current induction (eddy currents) in the various
conducting surfaces of the rest of the imaging
instrument. These, in turn, set up magnetic field
gradients that may persist after the primary gradients
are switched off. Such eddy currents are a problem in
both conventional and echo-planar imaging, but are more
severe in EPI. The gradient amplitudes, and particularly
the gradient switching rates, used in EPI are much
greater and induce larger eddy currents. Further, the
long readout period in EPI results in more opportunity
for image distortion from eddy currents.
A particularly common EPI artifact is so-called
"ghosting" from eddy currents. This results from the
time-dependent frequency shift created by the
time-dependent eddy currents. Because of the back and
forth trajectory in k-space used in EPI (see figure 3),
the frequency shifts create a phase difference from line
to line in the raw data. When the data are Fourier
transformed, the phase shift creates a phase ambiguity
in the images, such that part of the signal appears 90°
out of phase, or one half image away. This ghosting
structure is frequently referred to as an "N/2 ghost".
Figure 5 shows schematically the appearance of such a
ghost image.
|
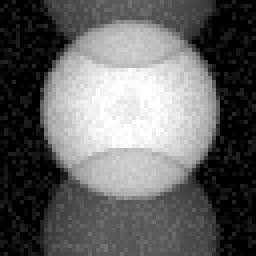 |
Figure 5. Appearance of a so-called N/2 ghost that
occurs frequently in echo-planar imaging. Such "ghosts"
are the result of small line-by-line phase errors that
can take place during spatial encoding.
The correction of image ghosts may take several forms.
Probably the most robust scheme is to design the
gradient coils critically such that eddy current
induction is minimized. At present the most effective
method is to use shielded or "screened" gradient coils,
in which a separate set of gradient coils is
counterwound around outside the primary coil set to
cancel any external magnetic fields. Because gradient efficiency
drops as the fifth power of the radius, even a small gap between
primary and secondary gradients allows for a non-zero gradient
inside the coil and a zero gradient outside.
Elimination
of the N/2 ghost also requires critical calibration of the
timing between signal digitization and gradient activity. Delays
of a few microseconds result in line-by-line phase discrepancies
because of the alternating left-right trajectory along the
readout axis in k-space (figure 3). These errors can be tuned
out in hardware by adjusting the sampling clock, and may be
adjusted further in software by adding an appropriate phase
shift to the raw data. The latter can be performed by an
exponential multiplication. An alternative approach to ghost
correction is to acquire a reference scan in the absence of
phase encoding and to use this as a basis for determination of
the time-dependent phase shifts.
5. Resolution
Resolution, or voxel size, depends on the maximum
gradient amplitude-time product in the raw data.
Increases in resolution require either increases in
gradient amplitude, increases in gradient duration, or
both. Neither is easy to come by. As we will see below,
the power requirements for the gradients in EPI can be
quite large. Further, switching to gradients
rapidly to very high amplitudes may ultimately result in
unacceptable safety problems. Increasing the duration of
the gradient pulses lowers the effective image bandwidth
and increases the sensitivity of the images to shape
distortion and other artifacts. When the
highest performance body gradient is set to reach
amplitudes of up to 3.6 gauss/cm with a rise time of 179 µsec
using a sinusoidal waveform. This results in a 3 mm pixel size
in the readout axis.
Fortunately, a variety of k-space encoding schemes are
available to improve spatial resolution. Increases in
resolution along the phase encoding axis are available
simply by extending the total duration of the echo
planar readout (box shown in dotted lines in figure 4). This increases the total displacement along the ky
(phase encoding k axis) at the cost of a decrease in
bandwidth and an increase in minimum echo time. Doubling
the encoding period, for example, will reduce the pixel
size and the bandwidth, per pixel, by a factor of two.
Shape distortions from field inhomogeneity will remain
constant in distance (as expressed in millimeters),
though they will cover double the number of pixels. This
tradeoff frequently works well.
A useful way to increase resolution along the readout
axis results from the Hermitian symmetry property of
k-space. Formally, reflections about the axes in
k-space are complex conjugates; a data point at (kx, ky)
is equal to the complex conjugate of the data point at
(-kx, ky) or at (kx, -ky). This symmetry property
implies that it is necessary to acquire only half of the
entire MR raw data space to form a complete MR image. A
very efficient way to achieve high resolution in a
single shot EPI experiment is to use a long readout
duration along ky and to acquire only the positive (or
negative) values in kx. Prior to image formation, it is
a relatively simple matter to calculate the data that
make up the uncollected portion of the image and then to
Fourier transform the entire raw data set to form a
complete image (figure 6A).
|
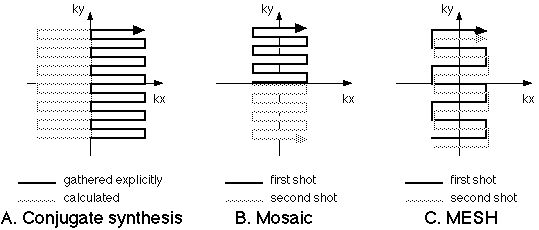 |
Figure 6. Resolution enhancement approaches for EPI. A.
The conjugate synthesis method (variously called
Half-Fourier, Half-NEX or partial-k) takes advantage of
the conjugate symmetry of k-space so that only half of
the raw data need be collected to form a complete MR
image. The Mosaic method (B) collects regions of k-space in
tiles. C. In MESH, the k-space regions are collected in
interleaved fashion, usually with a higher amplitude
phase encoding step.
The complex conjugation "trick" outlined above requires
a few pre-conditions to work properly. First, it depends
on the desired image having no "imaginary" component.
What this means in practice, is that the user must not
be interested in any phase deviations along the image.
Such phase difference might result, for example, from
local field inhomogeneities or motion and are usually of
little concern to the researcher in functional
neuroimaging. Secondly, any phase variations in the
image must be relatively small; otherwise, the
reconstruction will result in ghost-like artifacts from
locations with large phase shifts. Finally, the raw data
must be well-centered in k-space for the reflection
property to be accurate. Similarly to the process of
eddy current correction discussed above, this
requirement is achieved by a combination of hardware and
software engineering. Figure 7 shows example images,
each collected in a single shot, using the conjugate
synthesis approach. The in-plane resolution is about 1.5
mm.
|
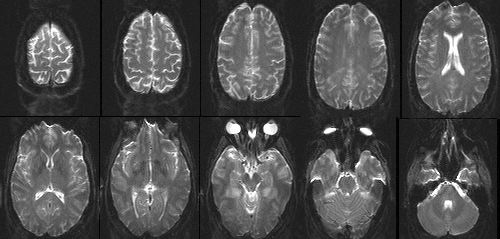 |
Figure 7. Single-shot EPI images of the human head with
1.5 mm in-plane resolution and 3 mm slice thickness,
collected using the conjugate synthesis method.
While true EPI is a single shot experiment (that is, a
single excitation pulse results in a complete image), a
variety of EPI hybrids, using multiple excitations per
image, may be used to increase spatial resolution. The
two most often used include the MESH and Mosaic methods. In the Mosaic method, figure 6B, each excitation
is used to cover a different region of k-space and the
resulting data are tiled together to form an image of
any desired resolution. The MESH technique is slightly
subtler. Here, a larger phase encoding step is used so
that data collected from separate RF excitations may be
interleaved. The important result in MESH imaging is a
wider bandwidth in the final image, which may be
desirable in reducing imaging artifacts. The multi-shot
hybrids have a signal to noise ratio advantage as well:
SNR increases with the square root of the acquisition
time, so that the SNR is about 40% better in the two
shot than in the single shot scans. The multiple shot
and conjugate synthesis methods can be combined over a
wide range of variations to produce echo-planar images
of very high resolution.
EPI resolution is limited, ultimately, by the SNR of the
images. Since typical EPI data are collected over 40 to
50 msec or so, as compared to the nearly 1 second of
acquisition time spent on a conventional scan, the SNR
is down by a factor of more than 4-fold at comparable
resolution on this basis alone. As shown in figure 7,
single shot images with voxel volumes of 3 x 1.5 x 1.5
mm (= 6.75mm3) offer acceptable SNR at 3 Tesla. In fact,
the measured SNR differences between conventional and
EPI data in human subjects are much less than predicted
on the basis of theory. This is likely due to the fact
that tiny motions in the conventional imaging set result
in an overall increase in apparent image noise. At the
present time, it seems that EPI spatial resolution is
largely gradient limited.
6. Hardware Requirements
EPI is a demanding sequence for the imaging instrument.
Good quality images require high performance gradients
with rapid rise times, high peak amplitudes, high
accuracy and low eddy currents. The demands on the data
acquisition system are considerable as well. Because the
data are sampled so rapidly, very fast analog to digital
converters (ADC’s), up to 2 MHz, are required. The ADC
subsystems, however, are becoming more widely available
due to advances in semiconductor technology.
6.1 Gradient Power
A typical body gradient coil in an MR system will have
an efficiency of about 1 gauss/cm per 100 amps. Thus, a
current of 250 to 350 amps is required to produce
acceptably high gradient amplitudes for EPI. Further,
the inductance of the typical body coil is about 1
milliHenry. The gradient slew rate (the rate of rise) is
determined by the rate of change in current (di/dt).
This is calculated easily. For example, to achieve a 200
µsec rise time to 3.5 gauss/cm requires that di/dt equal
350 amps/175 µsec, or 2 x 106 amps/second. With a coil
inductance, L, of 1 mH, the required driving voltage is:
V = Ldi/dt = 2000 Volts.
A conventional power system would need to deliver 2000
Volts at 350 amps, or 750,000 Watts to meet this
requirement. Most EPI-capable amplifier systems
therefore use some form of non-linear amplification
using either inductive or capacitive energy storage
devices. These implementations recognize that it is
generally not necessary to simultaneously source both
high current and high voltage to the highly reactive
gradient load.
The required accuracy of the gradient waveforms adds
another complicating factor. Any deviations from the
ideal waveform could result in phase errors in the
images. Such deviations can results from eddy currents,
physical instabilities or amplifier distortion. One
efficient way to manage this problem is to synchronize
the signal digitization to the integral of the measured
gradient activity, such that the data are sampled
uniformly in k-space
7. dB/dt and Safety and Head Gradients
It has been recognized since the early days of MRI that
the rapidly switched magnetic field gradients in imaging
instruments result in current induction in the patients.
Using the Hodgkin and Huxley equations for
neuronal excitability, suggests operational margins
below which gradient switching rate (dB/dt) was not
likely to be a cause of concern. At that time, gradient
switching rates above the predicted neural firing
threshold were not practical to achieve. Once the
non-linear amplifier methods became feasible, however,
it became possible to routinely exceed the threshold of
sensation using imaging gradients. Most of the
present day imaging systems have gradient performance
that is limited to just below the typical threshold of
sensory stimulation.
There is an alternative, however. The induced current in
the patient is proportional not only to the rate of
change of the magnetic field, but also to the
cross-sectional area of the body exposed to the changing
field. For example, it is now well-known that the
sensory threshold is higher for gradients that switch
along the sagittal axis than for those that switch along
the coronal axis, because the cross sectional area of
the typical supine person is much larger in the coronal
than in the sagittal plane. Further, the maximum dB/dt
occurs at the ends of the imaging coil, where the
magnetic fields are at their maximum. A shorter coil,
therefore, will have a reduced dB/dt. Probably the best
solution in functional neuroimaging is to build
head-only gradient coils. Such a devices gains
safety/sensory threshold margins due both to their
reduced length and to the reduced cross-sectional area
over which the gradients occur. The cross sectional area
of the human neck is less than one-sixth that of the
chest. The gradient coil length can be reduced by a
factor of at least 2, and probably more than three.
Therefore the stimulation threshold, in terms of imaging
gradient switching, will be reduced by at least ten, and
probably more than 20-fold.
Shorter and smaller gradients offer another set of
advantages: the gradient efficiency scales with fifth
power of the radius, so that reduced the diameter
two-fold can increase the gradient strength by a factor
of 32. Thus, the smaller gradients not only enable the
use of larger amplitudes with good safety margins, but
also make such strong gradients practical to implement.
At this writing, the major equipment manufacturers have
expressed little interest in special purpose head
gradients. In the end, however, it is likely the market
will demand such tools for high-performance functional
neuroimaging.
8. Contrast Variants
Because EPI is fundamentally just a spatial encoding
scheme, there are already a wide variety of variants
that can be used to offer a correspondingly wide range
of contrast behaviors.
8.1 Spin Echo
Figure 4 shows the most common implementation of EPI as
used for clinical imaging: the spin echo sequence. Here
the spatial encoding "module" is preceded by a 90°
excitation pulse and a 180° echo-forming pulse,
resulting in the formation of a Hahn echo during
the readout period. Such images have signal intensity
(SI) that is well described by the equation:
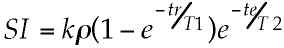
where k represents sequence independent factors such as
magnetic field strength and RF coil sensitivity, r is
the tissue proton density, tr is the repetition time and
te is the "echo time" or the time from the excitation
pulse to the center of the readout period. Such images
show relatively little sensitivity to local field
inhomogeneities (at least as they relate to contrast)
and behave similarly to conventional MR images. A key
difference, however, is that in a single shot EPI study,
the tr is effectively infinite, so that the images are
obtained with little or no T1 contrast. This is a
decided advantage in clinical T2-weighted studies where
the T1 and T2 contrast mechanisms when manifest
simultaneously, tend to result in an overall reduction
in image contrast.
8.2 Gradient Echo
The first practical rapid imaging method was arguably the FLASH
(fast low angle shot) sequence that has spawned a large number of
variants known collectively as "gradient echo"
techniques. The name refers to the fact that in these
sequences, the 180° echo-forming pulse is omitted, and
the signal is refocused solely by the gradients. EPI
versions of the FLASH scans are possible and are the
most commonly used method for functional neuroimaging
today. Figure 8 shows the generalized "gradient echo"
EPI sequence.
|
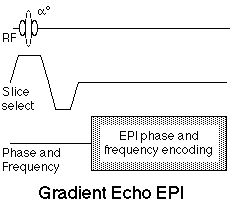 |
Figure 8. Gradient Echo EPI sequence. In this sequence,
the EPI data collection follows a single RF pulse, whose
flip angle, a, is adjusted to set up the preferred
contrast.
The gradient echo EPI sequence is used for several
reasons. First, and perhaps most importantly for
functional imaging, the contrast behavior includes a
T2*, as opposed to a T2 component. That is, the signal
intensity decays after excitation at a rate determined
by local field inhomogeneities. It is thought that the dominant
mechanism in so-called BOLD functional imaging is the
increased decay rate of the MR signal in the presence of
field inhomogeneities produced by deoxyhemoglobin. Thus,
an imaging sequence sensitive to such local variations
is ideal. In gradient echo EPI it is possible also to
use shorter TR’s without suffering large signal losses,
as the smaller excitation flip angle results in less
disturbance from magnetic equilibrium and therefore
shorter relaxation recovery times. Gradient echo EPI at
frame rates of up to 16 frames/second has been used to
produce good quality real-time images of the human heart
during the normal contractile cycle. For non-spin
echo scans it is possible to calculate the "Ernst"
angle, a, at which the signal will be maximal for any
combination of tr and T1:
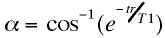
Since the ordinary functional imaging application is to
acquire a series of EPI scans at a non-infinite tr, the
gradient echo methods can confer a slight signal
advantage over spin echo studies. This advantage
increases at shorter tr.
8.3 Offset Spin Echo
By adjusting the relative timing of the Hahn spin echo
and the EPI readout module, it is possible to offset the
RF echo from the center of k-space. It is by now
well-known that the contrast in MR images is dominated
by the signal contrast that occurs at the center of
k-space, as this region of the raw data encodes the
largest spatial features of the images. In the offset
spin echo method, varying degrees of
susceptibility-related contrast are incorporated into
the images. This method has been suggested as an
approach to modulating somewhat independently the signal
loss from large and small field perturbers and as a
method for directly measuring line broadening. In
the limit, as the Hahn echo is delayed to beyond the EPI
readout, the offset spin echo scan becomes identical to
the gradient echo scan. The offset spin echo method may
have increasingly important applications at higher
magnetic field strengths where the inherent functional
imaging contrast is greater in BOLD studies and the
artifacts from non-ideal magnetic become more severe.
Figure 9 shows the offset spin echo sequence.
|
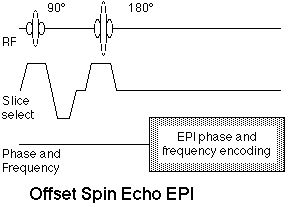 |
Figure 9. Offset spin echo EPI sequence. The offset
refers to the relative timing of the spin echo formed by
the 90°-180° pulse pair and the center of the EPI
readout period, which ordinarily is the center of
k-space.
8.4 Inversion Preparation
As discussed in the context of fat suppression, the
addition of a 180° inversion pulse prior to the standard
spin echo EPI sequence results in an inversion recovery
EPI scan. Such images have easily controlled T1 contrast
according to the equation:
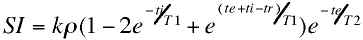
where
ti is the time between the inversion and
excitation pulses. The inversion recovery EPI sequence
is shown in figure 10.
|
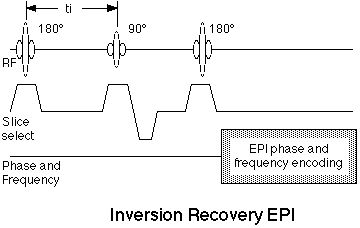 |
Figure 10. Inversion recovery EPI pulse sequence. The
addition of a 180° RF pulse at a time, ti, before the
standard spin echo EPI scan results in
inversion-recovery contrast behavior.
The inversion recovery method and its several variants
have been used to produced water-suppressed images,
similar to the FLAIR method used conventionally and have
proven useful in the measurement of blood flow and
perfusion.
9. Volume EPI
EPI as typically practiced is a two-dimensional encoding
strategy. In most cases, as suggested above, the third
dimension is provided by selective excitation. An
alternative is to use phase encoding in the slice
selection direction to create a 3D volume image,
as shown in figure 11.
|
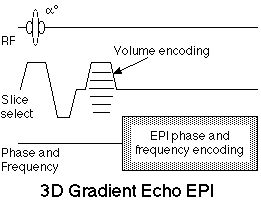 |
Figure 11. 3D, or Volume echo-planar scan. This is a
multi-shot EPI hybrid, where an additional phase
encoding step is applied along the slice selection axis
with each excitation. After 3D Fourier transformation,
these data yield a contiguous multi-slice image set. The
sequence is shown using gradient echo contrast, though a
180° echo-forming pulse can be added for spin echo
contrast.
Volume EPI has theoretical advantages in SNR. Because
the same volume is repeatedly sampled, albeit with
different phase encoding, the signal to noise ratio
scales with the square root of the number of phase
encodes, effectively the number of slices. Thus a 32
slice data set will have nearly six fold higher SNR than
a comparable single slice data set. Using a similar
volume sequence, it is possible to collect, for example
a sixteen slice volume study of the human heart in only
400 msec. This approach should be highly efficient
in functional neuroimaging as well, where contiguous
multi-slice image sets are highly desirable. The tr used
in the sequence must be reduced by a factor equal to the
number of slices to yield the same sampling density.
Thus, to acquire an image every 3.2 msec as part of a 32
slice volume, a tr of 100 msec is required. For this
reason, shallow flip angle, FLASH-type, imaging is used
so that the scanning can be performed at the tissue
Ernst angle.
This differs significantly from so-called echo-volumnar
imaging (EVI) in which the entire volume is collected
following a single excitation pulse. EVI is not entirely practical because the
extended total readout required greatly exceeds the
T2*’s of most tissues at clinical field strengths.
10. Conclusions
Echo-planar imaging is at this time the fastest and most
flexible approach to MR imaging, offering considerable
freedom in the selection of contrast and resolution
parameters. It is, however, a technologically
challenging method that requires that the imaging system
operate at near its performance limits in gradient
amplitude and rise times, system stability, and overall
noise figure. Further, EPI can suffer from serious
artifacts in shape distortion and image ghosts that
require extra attention from the researcher. All told,
however, the decided advantages of EPI in functional
neuroimaging have placed it very much in demand for fMRI
applications and have served to drive the technology
development both in the academic research laboratory and
with major commercial vendors, essentially all of whom
now offer EPI products.
Notwithstanding the considerable efforts that have gone
into EPI, there are still major advances to be gained.
Chief among them will be the practical performance gains
that can be achieved with ultra-high performance local
gradient coils, which will improve image quality by
reducing shape distortions and other bandwidth-related
artifacts, while increasing the comfortable operating
margin that avoids sensory stimulation.
With the relatively recent dissemination of product
level EPI hardware into consumer sites, the future of
EPI is rosy indeed and will continue to be driven by the
increasingly important clinical applications of
functional neuroimaging.
|
 |
|